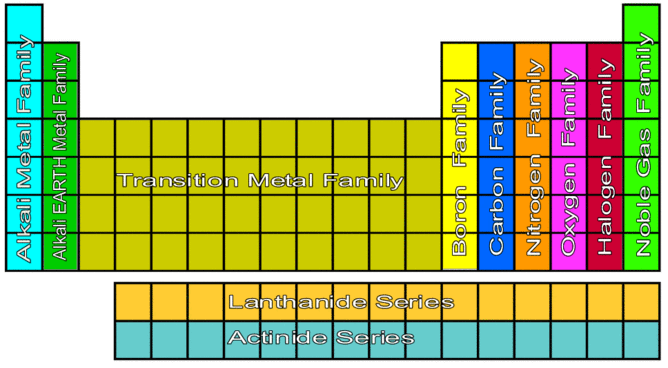
Alkaline earth metals are the Group 2 elements in the periodic table. What is the difference between alkali metals and alkaline earth metals?Īns. H can also gain an electron to form negatively charged ions, like the elements in the halogen group.It is also much less reactive, needing more energy for it to release that single electron to form positively charged ions.Despite having a single electron in its outermost shell, hydrogen (H) is not considered to be an alkali metal because: 
Why Is hydrogen not considered an alkali metal?Īns. Sodium and potassium are the most abundant alkali metals.ģ. What are the most abundant alkali metals?Īns. 
Being highly reactive, alkali metals in their pure form have to be stored in oil to keep them from coming in contact with air and water. Fr does not have any regular application as it is rarely present in nature. The top three elements of the group, Li, Na, and K, along with Cs, have different applications in various industries including glass-making, photography, weapons and explosives, and batteries Sodium compounds have some everyday uses in food processing and preparation. Video: Reactivity and Periodic Trends of the Alkali Metals Increasing reactivity (due to the increase in atomic radius, it is easier to knock out the valence electron in the elements located lower down the group, making Li the least reactive, and Francium the most reactive alkali metal).Increasing density and weight with Li being the lightest and Fr being the heaviest (though sodium and potassium are an exception as the latter is less dense than the former).Decreasing melting and boiling points.The lower down Group 1 on the periodic table you move, the following trends are apparent in the alkali metals: The heat resulting from the reaction ignites the hydrogen atoms produced, causing a considerable explosion. The final equation stands like the following:Įxamples of some other alkali metal-water reaction: The Na + and OH – ions are quite unstable in the aqueous solution, so they form sodium hydroxide. sodium (Na) comes in contact with water, the sole valence electron leaves the sodium atom and the reaction forms hydroxide and sodium ions along with hydrogen : These elements readily react with even the oxygen in the air to form oxides that make their shiny surface turn pale gray within moments if left in open air.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
